Current technologies to endotoxin detection and removal for biopharmaceutical purification - Schneier - 2020 - Biotechnology and Bioengineering - Wiley Online Library

PDF) Effect of peracetic acid–ultraviolet combination treatment on microbial and endotoxin levels in a pharmaceutical water system

Occurrence and fate of endotoxin activity at drinking water purification plants and healthcare facilities in Japan - ScienceDirect

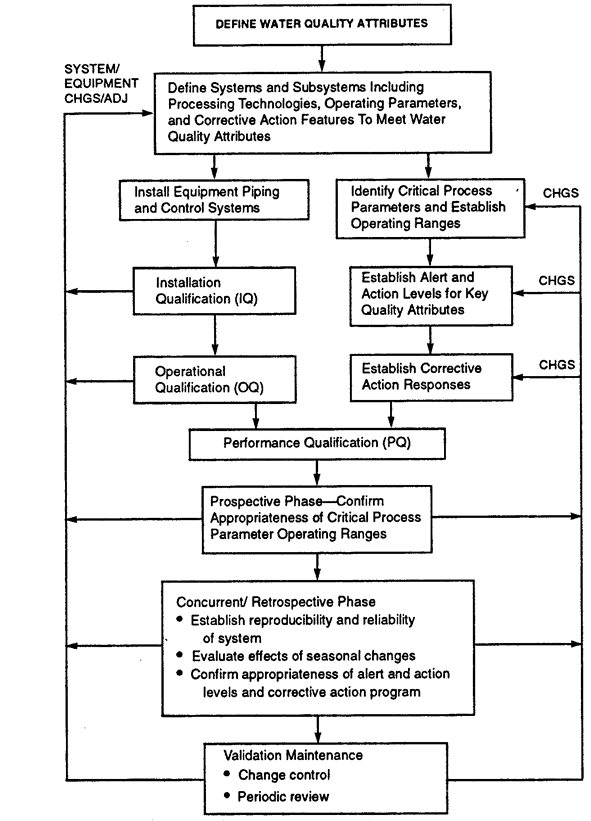
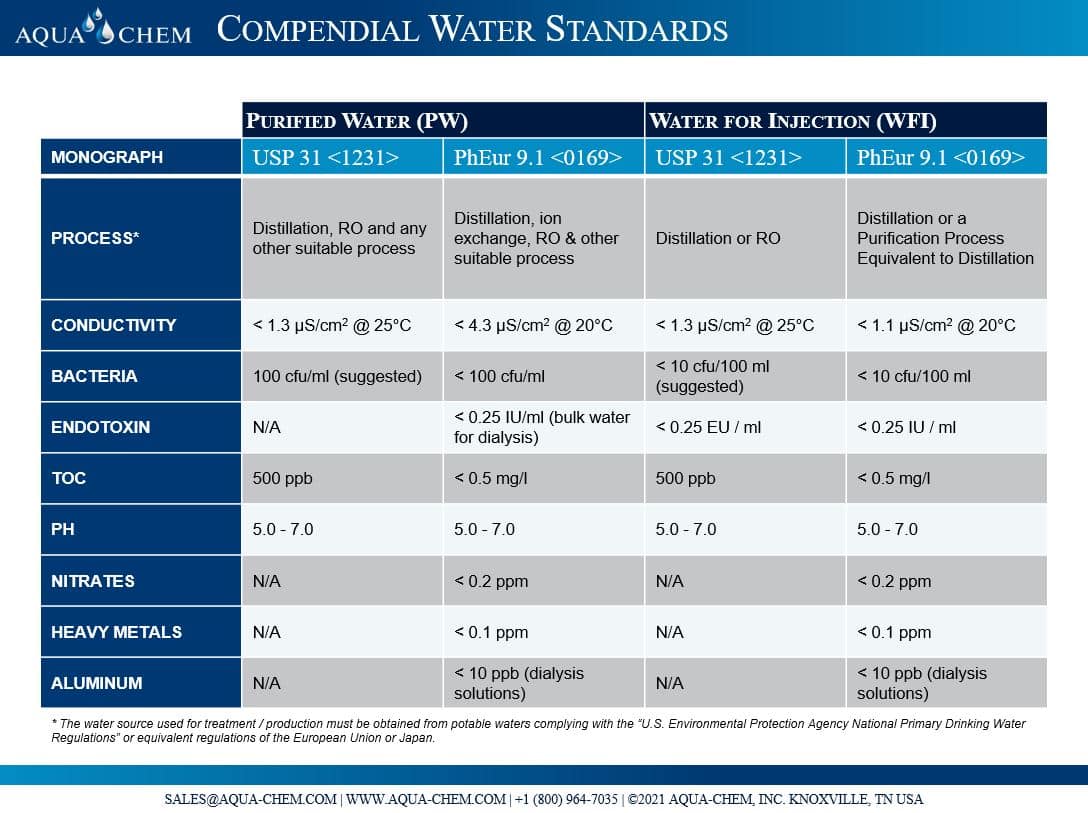
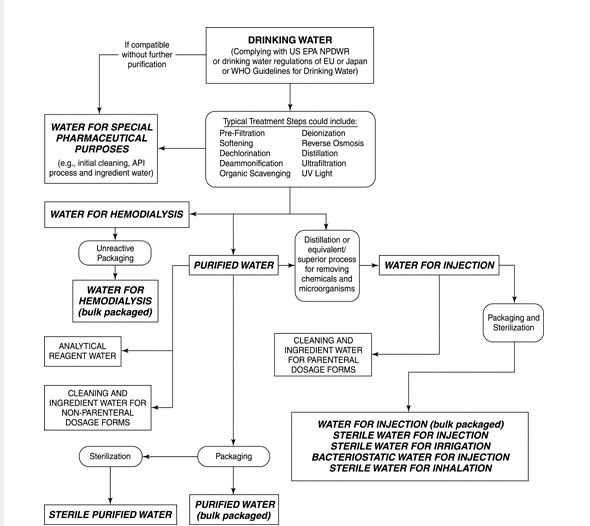
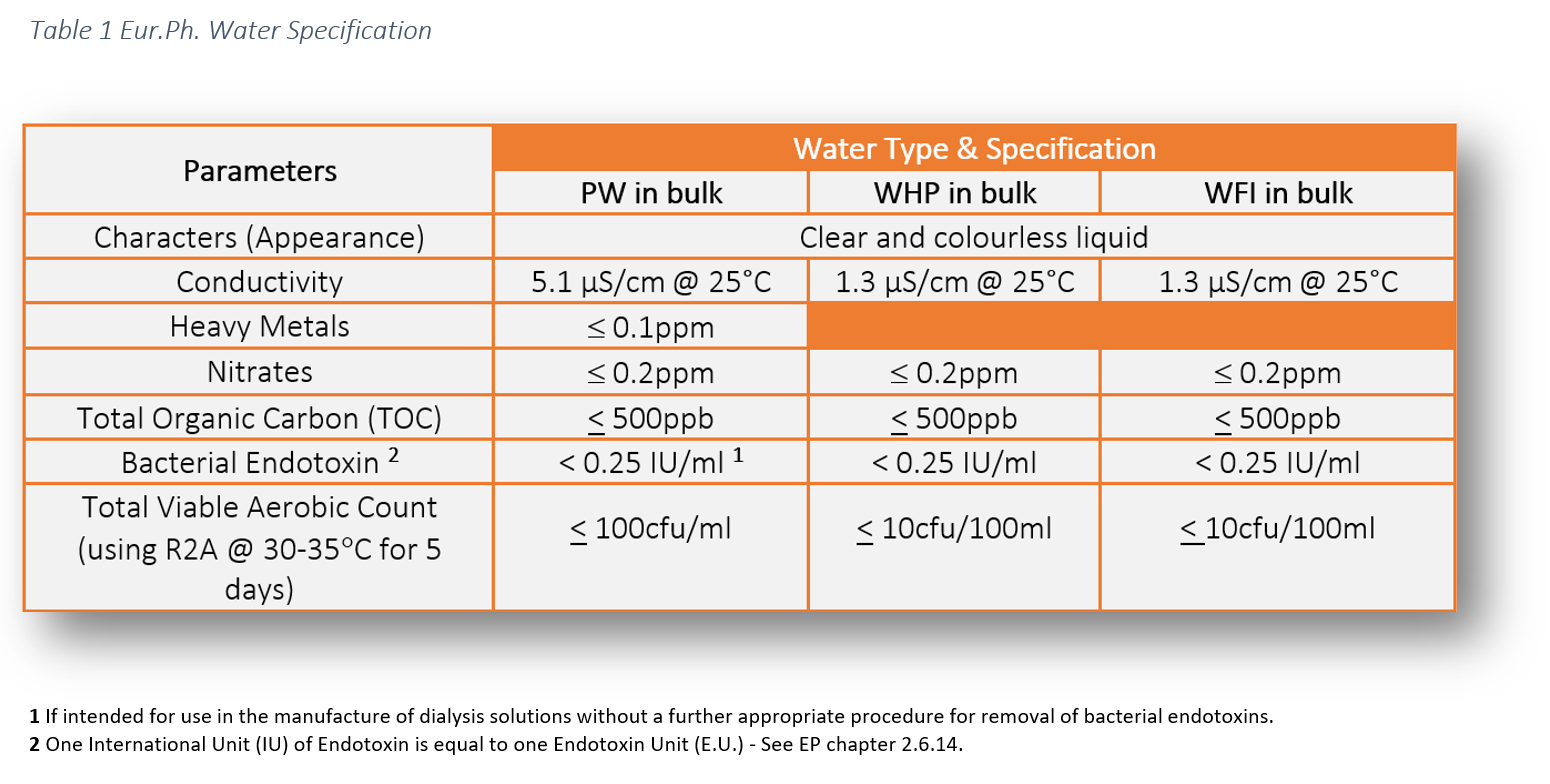
USP ; An Evolving Series of Informational Chapters on Depyrogenation | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
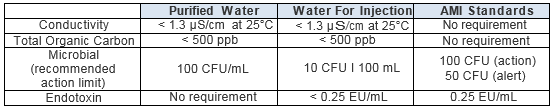

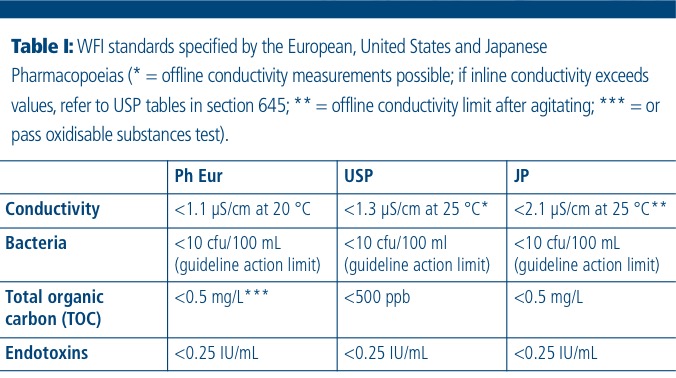
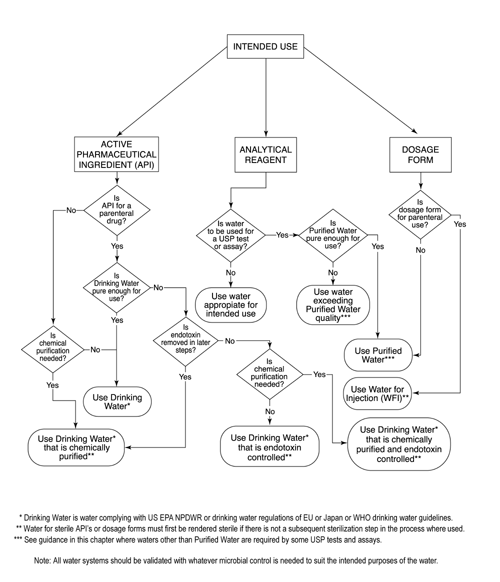
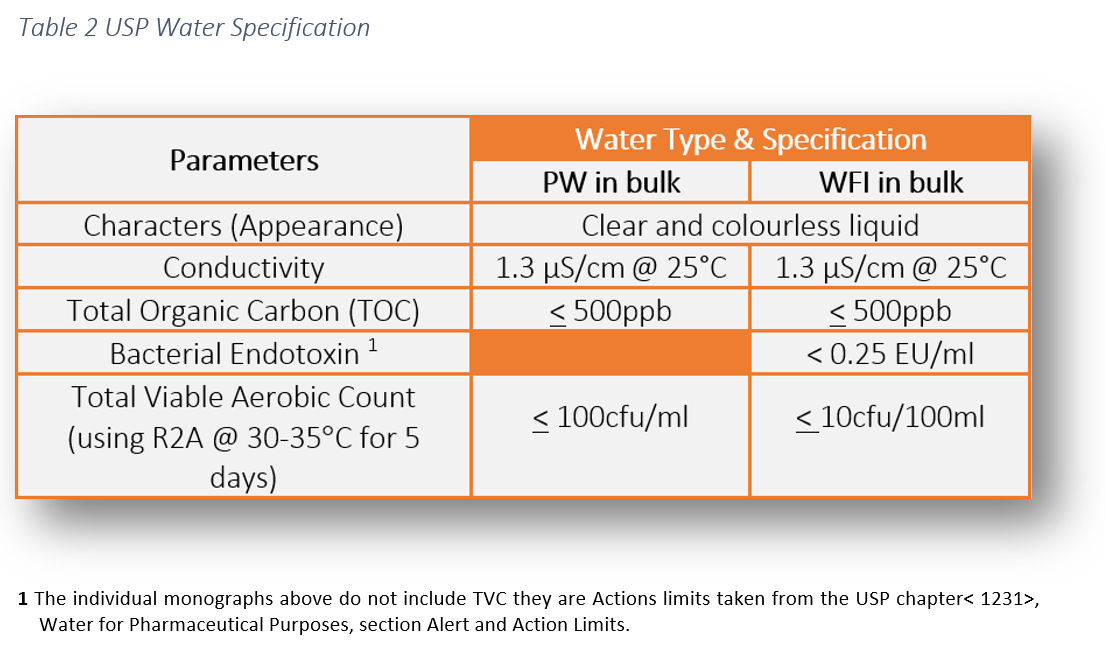
%20(5).png?width=600&name=Purified%20Water%20_%20Water%20for%20Injection%20System%20Design%20(Distillation)%20(5).png)